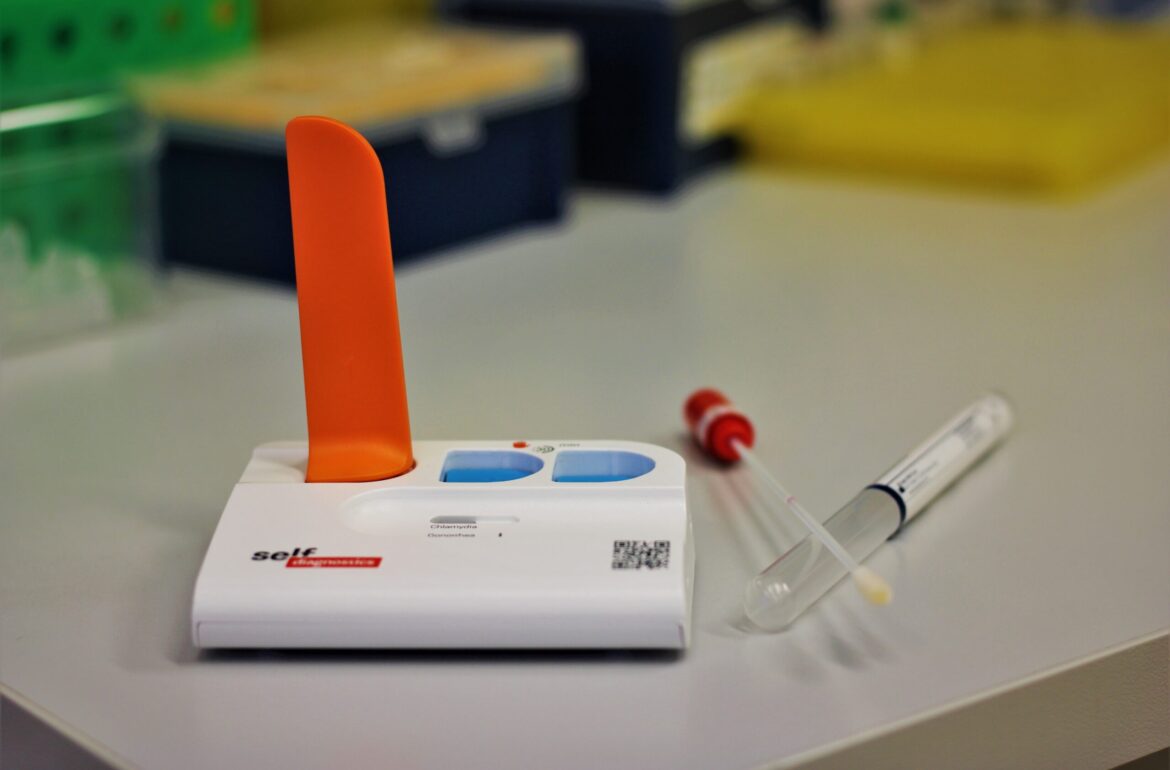
The University of Tartu and SelfDiagnostics have developed a SARS-CoV-2 rapid molecular test that allows detecting the viral RNA at a lower price and a greater resource efficiency. The test allows identifying infection on the spot in less than an hour.
The coronavirus pandemic has claimed over a million lives and the number of infected people is growing every day. This also creates the need for more testing and places a heavy burden on the central testing system.
The current method regularly used for virus identification is PCR, polymerase chain reaction, where the nucleic acid is amplified in stages at different temperatures. This method regularly presumes purification of the nucleic acids from the sample. The PCR method is, however, time-consuming and requires complex laboratory equipment, special facilities and trained staff. This makes current testing relatively expensive and resource-intensive. From the patient point of view, current centralised testing has two major drawbacks, significant delay in the results (usually days) and uncomfortable or even painful sampling of the nasopharynx.
University of Tartu researchers and Estonian-German company SelfDiagnostics have now developed a coronavirus rapid test that allows identifying the infection within 45 minutes. The test is based on the isothermal amplification of nucleic acids, meaning that the presence of viral RNA is determined at a constant temperature. This means that there is no need for large laboratory equipment or conditions and the test can be performed on-site or at the point of the patient care. Clinical studies indicate that the accuracy of the novel molecular rapid PCR test is similar to that of central laboratories.

According to Katrin Krõlov, the lead of the R&D activities, the test aims to get an accurate diagnostic result at the site of patient care. “Above all, the test is a faster and a more affordable alternative to current testing system,” added Krõlov. “For example, if you are travelling by plane, it is not very helpful if you are informed that you were on the same flight with a corona-positive person a couple of days after the flight. The rapid test, however, would allow identifying the infected people before boarding,” explained Krõlov.
Ülo Langel, Professor of Molecular Biotechnology, explained that the rapid test allows expanding testing in the regions where the fast diagnosing of the coronavirus is vital. Langel added: “The testing should be diffused and close to the patient. The bigger goal is to allow people test themselves without burdening the healthcare system. It would save valuable time in diagnosing fast spreading infection, where current testing system meets the limit.”
SelfDiagnostics has been developing different diagnostic products for the past ten years. According to its CEO Marko Lehes, they have successfully modified a previously developed infection rapid test to detect both the SARS-CoV-2 virus and influenza viruses. “The rapid test has the accuracy of a laboratory with the size of a human palm. We can say that it is basically a single-use mini-laboratory.”
The SARS-CoV-2 rapid test is developed in collaboration with the University of Tartu Institute of Technology and SelfDiagnostics. SARS-CoV-2 development is funded by EIT Health product development grant by the European Institute of Innovation and Technology. The molecular test platform development was supported by H2020, Sächsische AufbauBank, Archimedes and Enterprise Estonia.
For further inquiries please contact
Marko Lehes
CEO SelfDiagnostics
marko.lehes@selfdiagnostics.com
+372 52 151 05
Katrin Krõlov
Head of R&D, University of Tartu
katrin.krolov@ut.ee
 Back
Back