To make sure that antibiotics work, we need to know who is taking them and how. This data should be up in the cloud, an Estonian study concluded.
Every fifth baby is born via C-section. Many of these little humans would otherwise not be here now; modern medicine truly has saved countless lives. But it may all change soon, even within our lifetime.
Surgeries can often result in wound infections, which doctors typically prevent by administering antibiotics before and during surgeries. The spread of harmful bacteria could even be deadly otherwise. As University of Tartu professor Tanel Tenson explains, bacterial infections were the leading cause of death before antibiotics came to the market in the 1930s.
Tenson, who spent his life studying the matter, said that antibiotics could become obsolete, bringing us back to the pre-antiobiotcs era.
We already have examples of that. A bacterium called E. coli, a major cause of bloodstream infections in Europe, is resistant to antibiotics. Antibiotics are causing other problems too. Allergies, skin conditions and obesity have all been linked to excessive antibiotic use.
Tenson recently completed a study with his research group that sums up what is happening in Estonia and what we can learn from it, and those lessons can be applied anywhere. Similarly to other northern European countries, Estonia has a low antibiotic resistance level as this drug class is not exploited.
As the latest report by the European Centre for Disease Prevention and Control states, antibiotics resistance is the highest in the south and east of Europe and the lowest in the north of Europe.
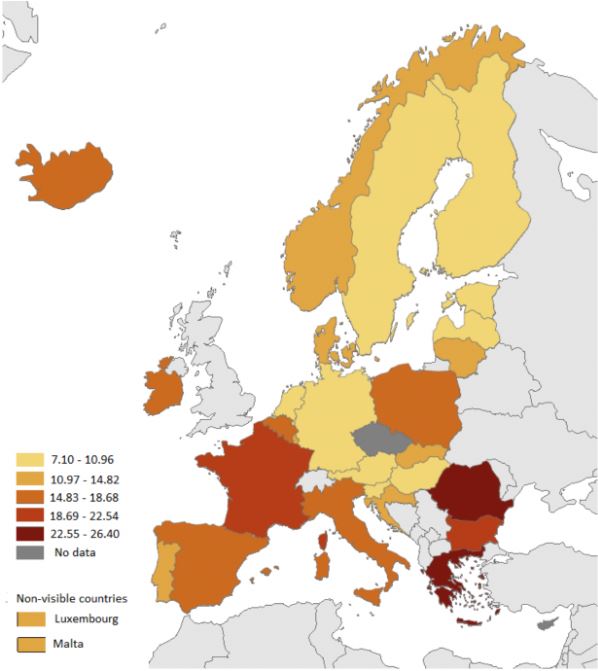
This doesn’t mean Estonia and its neighbours aren’t affected. As people and goods travel, the resistance spreads. The Estonian study led by Tenson showed that imported meat had a much higher concentration of resistant bacteria (for example E. coli) than local Estonian meat.
Clever bacteria
Antibiotics differ; some attack a wide range of bacteria, some only specific ones. Some are made naturally (Penicillin is made from mold), while others are manufactured by chemical modifications (the newest ones on the market). We inject them into pigs and chickens to prevent infections and promote growth. We swallow them in capsules or receive them in drips. However we take them, the antibiotics usually reach their resistance within 10 to 15 years, because bacteria, the first form of life on the planet, are clever. If something is threatening their existence (like antibiotics), they can mutate and even share the mutation with other bacteria, becoming a super bacterium.
We are all interconnected and our actions affect the ecosystem. Resistant bacteria and antibiotics are passed on via food and water. We only absorb part of the drug. Between 40 to 90 percent of the antibiotics remain intact and are expelled with urine and feces.
Even more concerningly, most antibiotics are now given to farm animals, from where they end up in the ecosystem through wastewater, compost, and slurry. Controlling and managing the use of antibiotics on farms is much more tricky. The study showed that there isn’t clear data on what type of antiobiotcs animals are given. Some farmers still prefer to make notes on paper, which is legal in most places.
It’s a hospital problem
Bacterial infections often occur in surgical sites and care homes since the elderly are generally more susceptible to various bacteria. According to the Estonian study, doctors don’t always find out what pathogen they are treating before prescribing antibiotics. It can lead to misuse and inappropriate use of the treatment.
The study also showed that smaller local hospitals prescribed antibiotics more freely than the major regional ones.

Medical centers that hired in-house infection control specialists had lower rates of antibiotic resistance. These specialists would monitor and manage the instructions for antibiotics use. Surprisingly, not all hospitals even have antimicrobial treatment guidelines that could reduce the relative risk for mortality by 35 percent, according to a Dutch study from 2016.
Tenson and his study group also found that doctors don’t always note down the reasons for prescribing antibiotics, don’t follow the instructions, or administer antibiotics when it’s not necessary. Even so, Tenson advises patients to follow the doctor’s recommendations. “Patients shouldn’t be expected to acquire enough knowledge to understand what the right thing to do is. It should be the doctors’ responsibility,” Tenson said.
What could we do?
Tenson believes that the key lies in data. “We want to create a full antibiotics resistance plan for Estonia, like nowhere else in the world,” he said. “If we were able to create a unified digital system that updates in real-time, then we could expand this system everywhere,” Tenson said.
He wants to gather antibiotics resistance test results and antibiotics given in real-time. There would be data drawn in from hospitals and farms. “Most of the data already exists in a digitized form. It’s just a matter of bringing all the systems together,” Tenson said.
This way, he explained, the experts could see where the resistance is at its highest. Based on the information, they would ask the hospital to use specific antibiotics and skip others. It would help keep the use of the antibiotic under control.
“Doctors shouldn’t just think of the one patient they are treating,” Tenson said. “Otherwise, they may heal one person, but then ten others would die because of the resistance.”
Minimizing the use of antiobiotcs is the key, Tenson said. On the farms, there should be better surveillance. Only certified vets with specific contracts should be able to prescribe antibiotics, to limit overuse.
In a way, Estonia and its lower than average antibiotics resistance is an example of how lifelong learning and informing doctors can make a difference, Tenson said.
It will be easier to adapt to this new bacterial pandemic since we all have the COVID-19 “training” behind us. This looming global outbreak is simply about thinking of others around us who may get infected. This time, we could be having a powerful global data tool to back us up.
Written by: Marian Männi
This article was funded by the European Regional Development Fund through Estonian Research Council.
 Back
Back



